
Hubin’s Group Develops Oxidation Catalysts to Purify Wastewater
EPSCoR Update - May 2023
Wastewater containing contaminants of emerging concern (CECs) including organic dyes pose a potential threat to the aquatic ecosystems as well as living organisms and humans. Organic dyes have been widely used for industrial and commercial purposes. Many organic dyes are potentially toxic and carcinogenic in nature. To address this environmental and water re-use concern, it is important to degrade dyes as well as other organic pollutants or CECs.
Elimination of the CECs from wastewater is environmentally necessary, as well as being necessary for the acceptance of purified water for reuse by humans. Conventional methods for treating CECs include usage of iron-based metal catalysts and photocatalysts involving semiconductor metal oxide nanoparticles. However, these conventional catalysts suffer from several drawbacks such as stability in a narrow pH range, low surface area, high cost, poor scalability, and limited ability to absorb visible light.
Hence, Dr. Timothy Hubin, Department of Chemistry and Physics Chair, Southwestern Oklahoma State University (SWOSU), collaborated with Drs. Somrita Mondal (SWOSU), Timothy Prior (University of Hull, Kingston Upon Hull, UK), Allen Oliver (University of Notre Dame, IN), and Jeanette Krause (University of Cincinnati, OH), and SWOSU students : Leslie Garcia, Tuyet Hoang, Jessica Thompson, and Michael Allen to design transition metal- cross-bridged tetraazamacrocycle oxidation catalysts for degradation ofCECs including organic dyes leading to waste water purification.


Hubin’s group has developed a series of oxidation catalysts, which can effectively bleach organic dyes at room temperature over a wide range of pH. The research team have successfully synthesized and characterized a wide variety of ethylene cross-bridged tetraazamacrocyclic ligands with different functional groups and their transition metal complexes, which can act as oxidation catalysts.
“Our cross bridged tetraazamacrocycles are highly stable in wide pH range, are economically affordable, and are capable of bleaching organic dyes at room temperature and normal light,” Hubin said.
“Our catalysts are scalable, and can be synthesized in large scale, which is an essential condition for treating wastewater in large water bodies,” Mondal added. The research team have monitored the catalysts’ capability to bleach three dyes (~ 1 mM concentration): methylene blue (MB), methyl orange (MO) and Rhodamine B (RhB).
“Our catalysts have displayed maximum efficiency in bleaching of Methyl Orange (MO) resulting in a very high turnover frequency (TOF up to 175/hour), which confirms their excellence in catalytic activity, but our catalysts have been least effective in degrading Rhodamine B (RhB) among the three dyes we have explored so far,” Hubin said.
“The highest turnover frequency (TOF) our catalysts can achieve with the bleaching of RhB is ~ 13, which is a quite satisfactory value for catalytic activity,” Mondal added.
The current research from Hubin’s group can pave the way for successful design of a series of oxidation catalysts for degradation of dyes and CECs leading to wastewater purification. Soon, the research team plans to examine different metals and ligands to design a wide variety of catalysts for better efficiency in dye bleaching, to embed the catalysts into polymer support to make them heterogeneous for recyclable use of catalysts, and to explore the use of the catalysts for degradation of different CECs other than dyes. The team also wants to explore the real-life application of the catalysts they developed for purification of wastewater in water bodies.
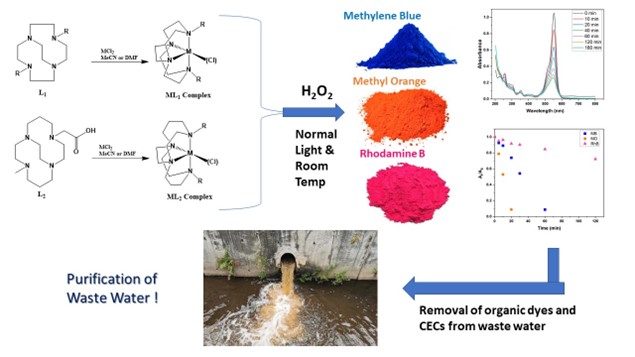
Synthesized metal-ligand tetraazamacrocycles can significantly bleach organic dyes Methylene blue, Methyl orange and Rhodamine B at room temperature and normal light in presence of hydrogen peroxide, leading to wastewater purification from harmful dyes and CECs
____________
Funding for this project was provided by the National Science Foundation under Grant No. OIA-1946093 through OK NSF EPSCoR
